8 dez 2017 ano - SaMD Clinical Evaluation Draft Guidance
Descrição:
The draft guidance pertains to the conduct of clinical evaluation of Software as a Medical Device (SaMD) and focuses on the general principles of clinical evaluation, which includes establishing the scientific validity, clinical performance, and analytical validity for a SaMD. This draft guidance is not final nor is it in effect at this time.
Adicionado na linha do tempo:
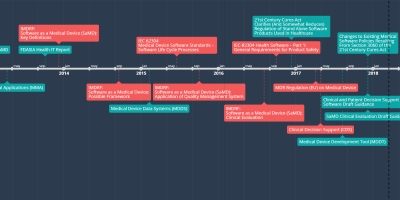
SAMD Timeline
Data:
