8 dic 2017 año - Changes to Existing Medical
Software Policies Resulting
From Section 3060 of the
21st Century Cures Act
Descripción:
A draft guidance that provides clarity on FDA’s current thinking regarding changes made by the 21st Century Cures Act (Cures Act) to the definition of a medical device and the resulting effect on guidances related to medical device software. This draft guidance is not final nor is it in effect at this time.
Añadido al timeline:
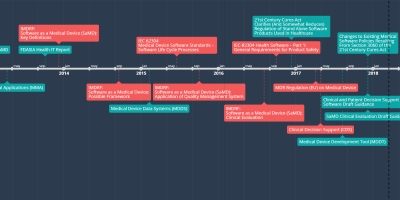
SAMD Timeline
fecha:
