sep 18, 2014 - IMDRF:
Software as a Medical Device:
Possible Framework
Description:
This document introduces a foundational approach, harmonized vocabulary and general and specific considerations, for manufacturers, regulators, and users alike to address the unique challenges associated with the use of SaMD by establishing common vocabulary and an approach for categorizing SaMD, identifying specific information provided by the SaMD, and providing criteria to categorize SaMD based on the combination of the significance of the information provided by the SaMD.
Added to timeline:
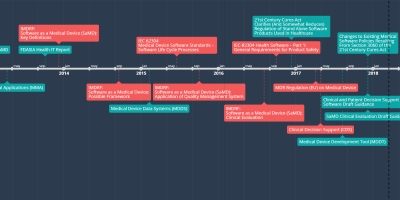
SAMD Timeline
Date:
