jan 1, 1911 - Ernest Rutherford
Description:
He thought atoms were made up of empty space where electrons orbited. He thought hypothetically atoms had a small, central, positively charged nucleus. He conducted an experiment in which he could figure out atoms had empty spaces to be able to let particles pass through solid objects, mostly. He also found out that atoms must have a part in them dense enogh to stop particals from going through.
Added to timeline:
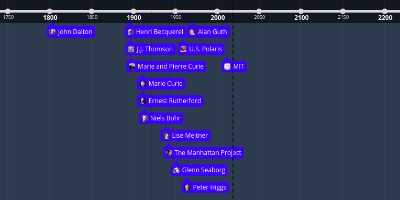
Atom timeline
Date:
Images:
![]()
