Brenna Fleming: timeline
Category: Other
Updated: 8 Dec 2017
Contributors

Created byBrenna Fleming
Attachments
Events
a total of 63 elements had been discovered
up until this pointIn 1817 Johann Dobereiner noticed that the atomic weight
of strontium fell midway between the weights
of calcium and barium, elements
possessing similar chemical properties.The law of Triad was createdBetween 1829 and 1858 a number of scientists found that these
types of chemical relationships extended beyond the triad.John Newlands, an English chemist, wrote a paper in 1863 which
classified the 56 established elements into 11 groups based
on similar physical propertiesMendeleev's table became available to the scientific
community via publication in 1869 before
Meyer's appearedMendeleev developed his statement of the periodic
law and published his work On the Relationship
of the Properties of the Elements to their Atomic Weights in 1869.In 1906, Mendeleev came within one
vote of being awarded the Nobel Prize for his work.Lord Rayleigh reported the discovery of a new gaseous element named argon which proved to
be chemically inert.Ernest Rutherford published studies of the scattering of alpha particles by heavy atom nuclei
which led to the determination of nuclear charge.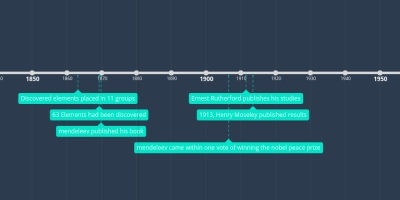
Comments