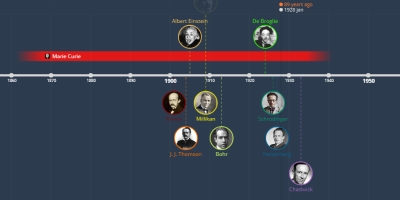
jan 1, 1913 - Bohr
Description:
Electrons move around the nucleus in fixed orbits, an electron in a particular orbit has constant energy. An electron can absorb energy and move to a higher energy orbit of larger radius. An excited electron can fall back to its original orbit by emitting energy as radiation. Electrons can only exist in certain discrete energy levels.Added to timeline:
Date:
jan 1, 1913
Now
~ 112 years ago