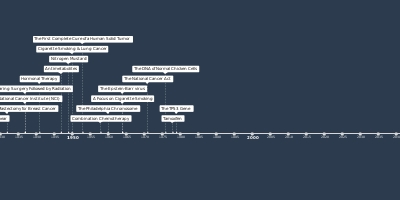
1 gen 1997 anni - Rituximab
Descrizione:
For use in patients with treatment-resistant, low-grade or follicular B-cell non-Hodgkin lymphoma (NHL), the Food and Drug Administration (FDA) approves rituximab, a monoclonal antibody.Aggiunto al nastro di tempo:
Data:
1 gen 1997 anni
Adesso
~ 28 years ago