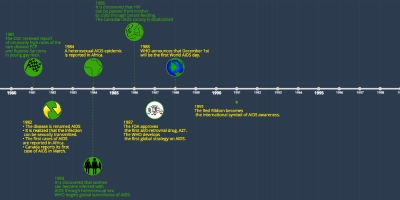
5 ene 2016 año - 2016 Health Canada approves use of daily oral Truvada (tenofovir plus FTC) for use as pre- exposure prophylaxis (PrEP) to reduce the risk of sexual transmission of HIV. Updated results from the HPTN 052 and PARTNER studies continue to show that antiretroviral treatment (ART) and an undetectable viral load significantly reduces the risk for HIV transmission through both anal and vaginal sex
Añadido al timeline:
fecha:
5 ene 2016 año
Ahora mismo
~ 8 years and 4 months ago