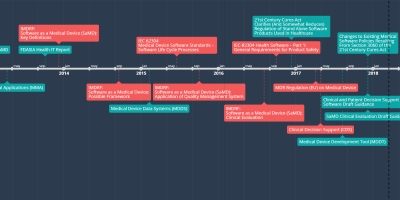
5 abr 2017 ano - MDR Regulation (EU) on Medical Device
Descrição:
The regulation provides rules concerning the placing on the market, making available on the market or putting into service of medical devices for human use and accessories for such devices in the Union.
Adicionado na linha do tempo:
SAMD Timeline
Data:
