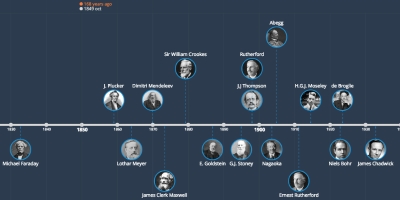
6 mar 1911 ano - Ernest Rutherford
Descrição:
Using alpha particles as atomic bullets, probed the atoms in a piece of thin (0.00006 cm) gold foil . He established that the nucleus was: very dense,very small and positively charged. He also assumed that the electrons were located outside the nucleus.Adicionado na linha do tempo:
Data:
6 mar 1911 ano
Agora
~ 114 years ago